The family of an Anaheim teacher, who recently passed away, spoke out regarding bone material being used in medical surgeries.
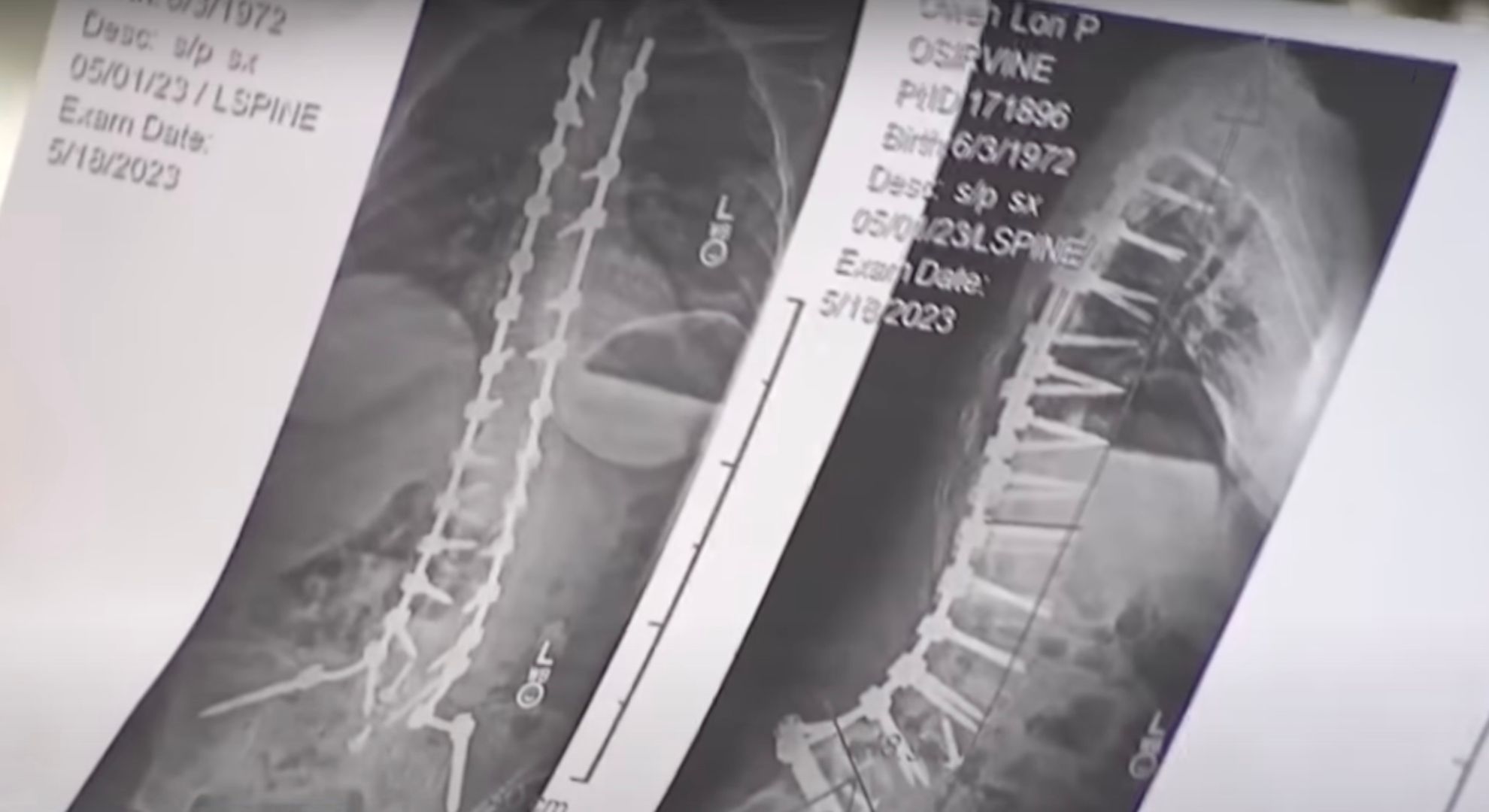
Earlier this year, Lori Peterson Owen underwent a 9-hour back surgery for scoliosis at the Hoag Orthopedic Institute in Irvine. Having stable vital signs, she was sent home. As she was about to celebrate the success of her surgery, she suddenly fell very ill several days later.

When she began acting differently, they decided to call 911 for help, and they were sent to the UC Irvine Medical Center. Lori was hooked onto life support, and doctors found out that she had Tubercular Meningitis.
Tubercular Meningitis refers to the swelling around the brain. This is the germ that causes tuberculosis.
During her said surgery, her screws were held in place with the help of a product called “ViBone” manufactured by Aziyo Biogenics.
It took being admitted to UC Irvine to find out that the product had been the cause of her contracting Tubercular Meningitis.
Matt Zahn, MD, said, “Presumably, the donor of for whom the tissue material came was infected with tuberculosis.”
Lori passed away just days before the pharmaceutical company issued a recall on the product. According to a report by the Centers for Disease Control and Prevention (CDC), the contaminated batch was sent to 13 surgical and dental clinics in seven states in America. It was also reported that at least two patients died upon receiving the product this year.
A similar scenario occurred back in 2021 in Indiana, but it involved a different bone-repair product from the same manufacturer, resulting in the deaths of eight patients.
The doctors who spoke to Lori’s family presented them with two options: either they let her go, or she will be confined to a skilled nursing facility.
They then decided to give her up and came to publicize what happened to their daughter to spread awareness.
Aziyo Biogenics has since rebranded to Elutia. They have not yet released a statement regarding this issue. The company has said that they have previously tested the ViBone Lori received and tested negative for tuberculosis. Federal regulations state that it is not required to test bone tissue donors for TB or TB risk factors.
Other POP! stories that you might like:
AI companies are looking for authors to improve their AI-generated ‘creative writing’
Filipino internet sensation Sammy Manese passes away
Grieving father of Swiftie who died in Brazil seeks answers from ‘Eras Tour’ organizers
Foreign YouTuber shows tricycle driver’s scam of charging P550 for a 600-meter ride
‘AI Artist?’: Student from Antique earns mixed reactions for his creations made using AI